
Winter, " Chemical Bonding", Oxford University Press, 2nd edition, 2016, ISBN: 978-0-19-870095-1.Ĭhemputer, a set of simple calculators for chemistry: Ĭopyright 1993-2021 Prof Mark J. Being mutually negatively charged, the electron pairs repel the other electron pairs and attempt to move. It’s useful to study molecular geometry to get information beyond that provided in a Lewis structure. Electron-pair Geometry versus Molecular Structure. 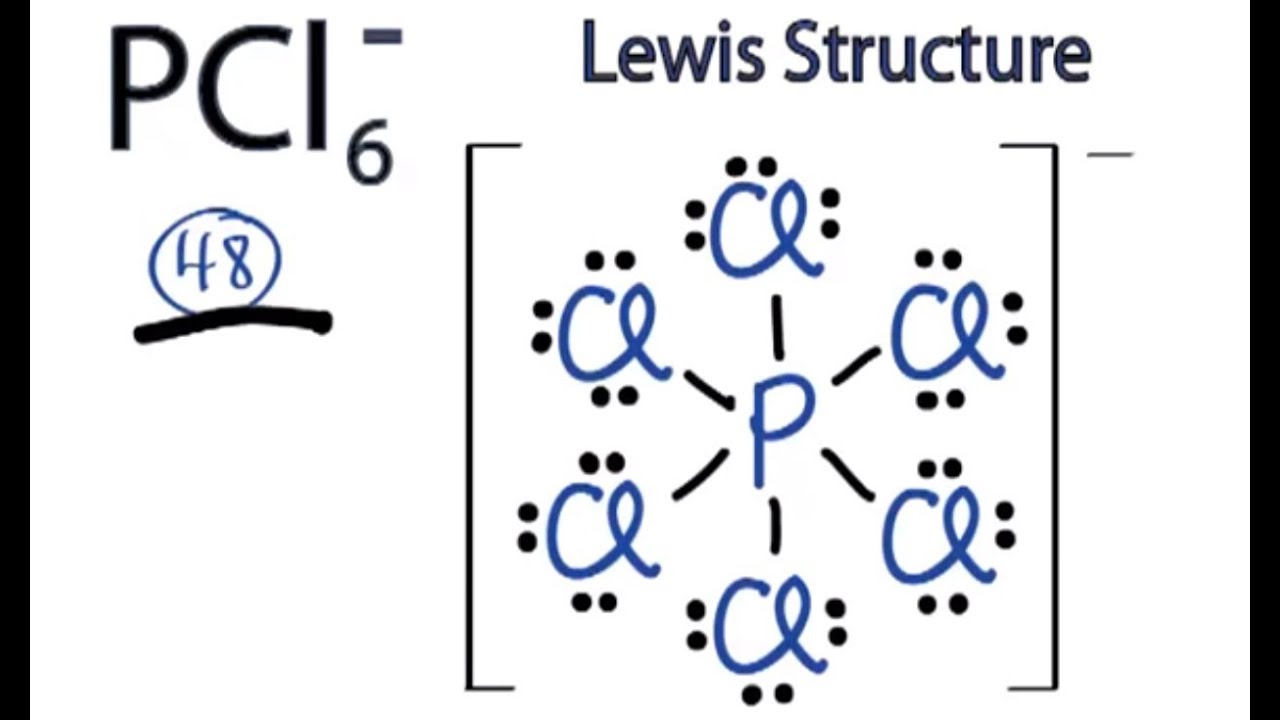
The VSEPR calculation used here is described in detail within the Oxford Primer: Mark J. Molecular geometry is a method to determine the shape of a molecule based on the repulsion occurring between bond electron pairs in the outermost (or valence) electron shell. Molecules can then be divided into two groups: Group 1: Molecules with NO lone electron pairs. Q-Chem begins a calculation by taking the user-defined coordinates and. Electron pair geometry is determined from the total electron pairs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
